Aging Isn't One Clock. It's Many
New Stanford research reveals your organs age at completely different speeds — and a blood test can now tell which one is running out of time.
Two people can both be 50 years old.
But one might have a heart that functions as if it belongs to a 65-year-old, while their brain still runs like that of someone in their early 40s. The other person might have the opposite problem entirely.
We’ve always thought of aging as something that happens to the whole body at roughly the same pace. You get older, and everything gets older with you.
New science says that’s wrong — and the implications for how we detect and prevent disease could be significant.
But more importantly, in many cases, the organ that ages fastest is the one most likely to develop disease later.
Waves of Change: How Aging Accelerates at Key Ages You Wouldn’t Expect
Before we get into organs, there’s a 2019 finding that didn’t get nearly enough attention.
Researchers at Stanford analyzed blood samples from hundreds of people across different ages and found that aging doesn’t just creep along steadily. It appears to happen in distinct waves, with three major biological turning points hitting at around ages 34, 60, and 78.
At each of these ages, a large number of proteins in the blood show sudden, noticeable changes. The body doesn’t just quietly age. It shifts.
This was surprising because 34 feels young. Most people at that age don’t think of themselves as “aging.” But biologically, something changes — the first of several inflection points in how the body operates.
That study planted an important seed: Aging is more complex and more uneven than we assumed.
What came next pushed that idea much further.
Your Body’s Secret Clocks: Why Organs Age on Their Own Timelines
In 2023, the same Stanford researchers published a landmark study in Nature that took this idea to a new level.
They analyzed blood samples from roughly 1,400 people, measuring nearly 5,000 different proteins circulating in the bloodstream. Then they validated what they found by testing the approach on another 4,000 people.
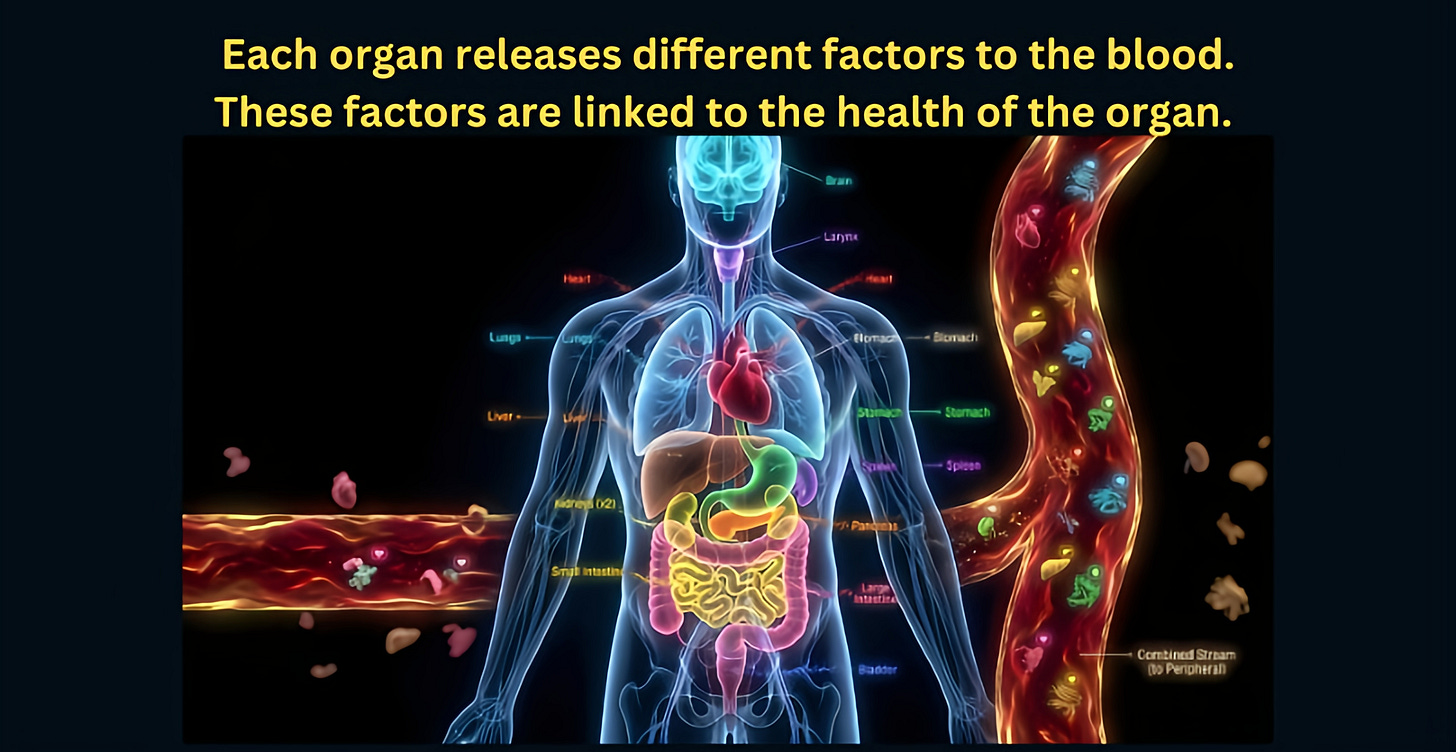
Here’s the key insight: many of these proteins don’t come from just anywhere in the body. Specific organs release specific proteins into the blood as part of their normal daily activity. The heart releases certain proteins. The kidneys release others. The brain releases its own distinct set.
By tracking which proteins came from which organs, the researchers were able to estimate something remarkable: the biological age of 11 different organs and systems in the body, including the brain, heart, liver, kidneys, lungs, pancreas, muscles, immune system, arteries, fat tissue, and intestines.
And what they found was striking.
About one in five adults had at least one organ aging significantly faster than you’d expect for their age. A 50-year-old might have kidneys functioning like a 60-year-old’s, while their heart appeared younger than average.
In other words, your body doesn’t run on one clock. It runs on many — and they don’t all keep the same time.
Speeding Clocks = Higher Risks: Why Accelerated Aging Spells Trouble
Interesting as it is to know your organs age at different speeds, the most important finding was what that difference meant.
When researchers followed participants over time, accelerated organ aging turned out to predict disease risk — often years before any symptoms appeared.
A biologically older heart was linked to higher rates of heart failure and cardiovascular disease. A faster-aging kidney predicted metabolic disease and high blood pressure. A biologically older brain was associated with cognitive decline and a higher likelihood of dementia.
Even relatively small differences mattered. People who had even one organ aging significantly faster than their peers faced a 15% to 50% higher risk of dying over the following 15 years, depending on which organ was affected. People with two or more rapidly aging organs faced risks up to six times higher than those with none.
This is the shift that aging researchers have been working toward for decades: moving from detecting disease after damage is already done to identifying which organs are under stress before anything goes wrong.
The Power Duo: Brain and Immune Systems as Aging’s Master Keys
A large follow-up study published in Nature Medicine in 2025 pushed the question further. This wasn’t a small replication effort — it analyzed data from nearly 45,000 people in the UK Biobank, one of the largest biological research databases in the world.
The researchers asked: across all 11 organ systems, which ones most strongly predict how long and how well you’ll live?
Two stood out above all the others.
The brain.
And the immune system.
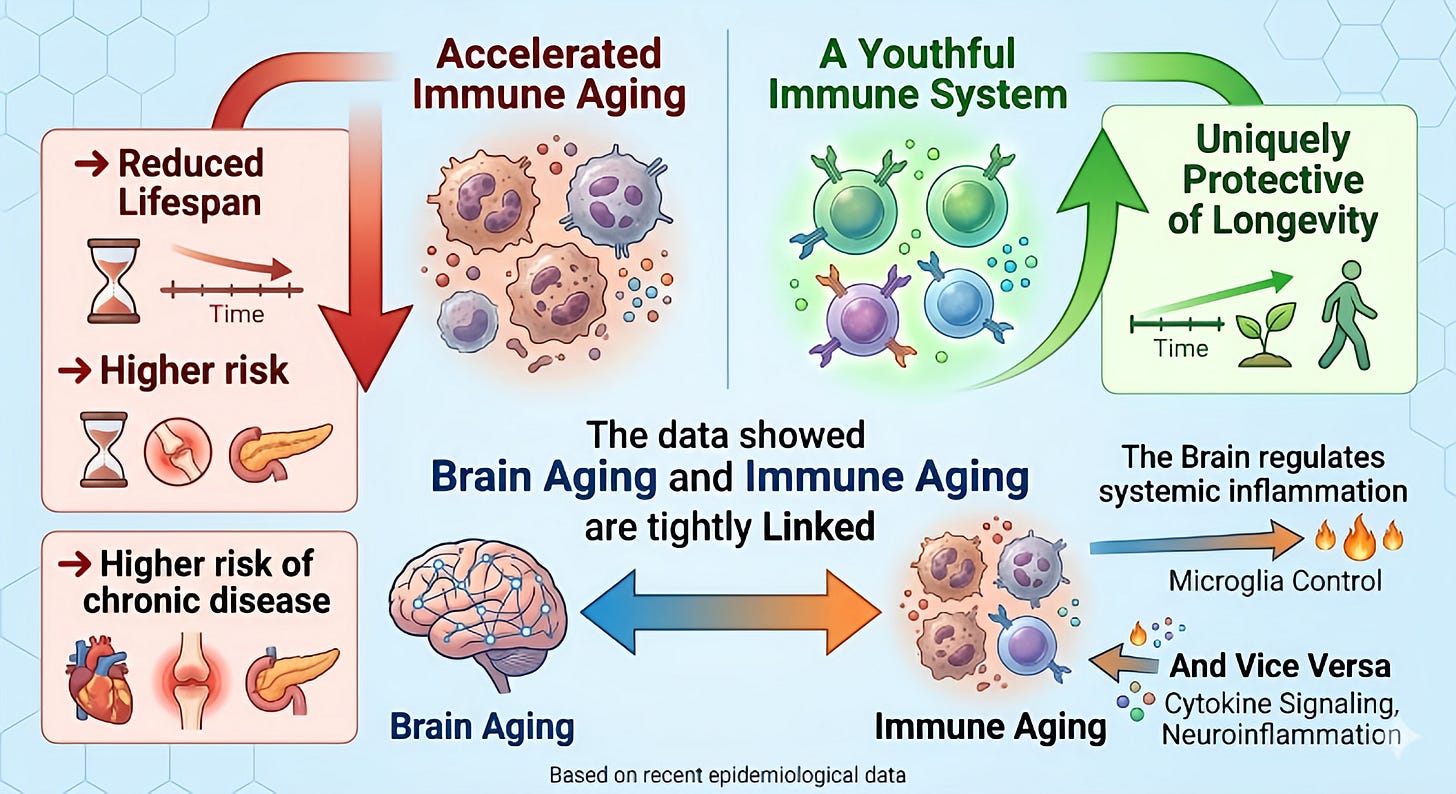
People whose blood proteins suggested accelerated aging in those two systems were significantly more likely to experience chronic disease, physical decline, and earlier death — regardless of what their other organs were doing.
This fits with what biology has been telling us in other ways.
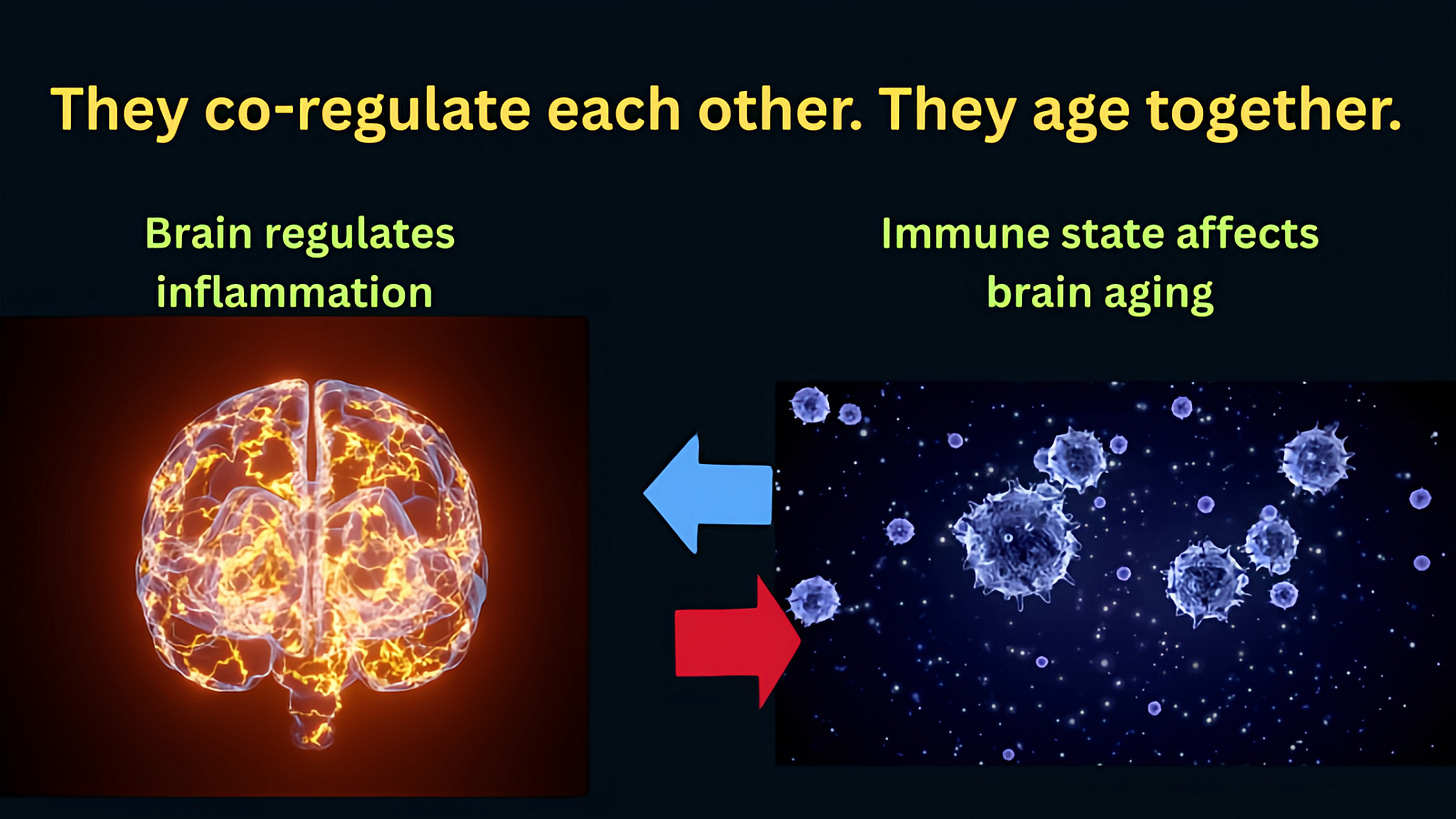
The brain regulates almost everything: metabolism, stress hormones, sleep, and behavior. When it starts to decline, effects ripple outward to the whole body. The immune system, meanwhile, is the body’s master maintenance crew. It clears out damaged cells, manages inflammation, and repairs tissues. When it ages, inflammation rises — and sustained inflammation underlies nearly every major chronic disease, from heart disease to Alzheimer’s to cancer.
These two systems may sit closer to the center of aging than we previously understood. When they go, a lot else tends to follow.
Beyond the Barrier: Why Brain Signals Appear in Blood Tests
This might be the part that’s hardest to believe. The brain is protected by something called the blood-brain barrier, a tightly controlled filter that limits what can pass between the brain and the rest of the body. So how can a blood sample tell us anything about brain aging?
It turns out the brain is not as isolated as we once thought.
Brain cells and the blood vessels running through them constantly release proteins during normal activity, and even more so under stress. Small fragments of those molecules do make it into circulation, and they carry information about what’s happening up there.
When researchers looked at which proteins most strongly signaled accelerated brain aging, they found them clustered around processes central to brain disease: inflammation, disrupted blood flow, changes in how neurons communicate, and breakdown of the structural scaffolding that holds brain tissue together.
These are the same processes that drive Alzheimer’s disease.
Here is where the numbers get striking. Having an especially aged brain was associated with a 3.1 times higher risk of developing Alzheimer’s disease, roughly the same as carrying one copy of APOE4, which is the strongest known genetic risk factor for the condition. On the flip side, having a biologically youthful brain was linked to a 74% lower risk, a level of protection comparable to carrying two copies of the protective APOE2 gene variant.
Put together, people with aged brains had a 13.5 times greater risk of developing Alzheimer’s compared to those with youthful brains.
To put that in context: APOE4 is the gene variant that people get tested for when they want to understand their Alzheimer’s risk. It is the gold standard of genetic risk factors for the disease. The fact that brain aging detected through a blood test carries comparable predictive power is a significant finding.
And crucially, these associations held up independently of age, sex, and a person’s APOE genotype, Stanford Medicine, meaning this is not simply measuring the same thing as genetic testing. It is capturing something additional and separate.
Alzheimer’s currently has no cure, and by the time someone receives a diagnosis, significant brain damage has typically already occurred, often after years of silent deterioration. The disease does not announce itself. What this research suggests is that the molecular warning signs could be visible in a routine blood draw years or even decades before a single symptom appears. That would represent one of the most consequential shifts in dementia medicine in a very long time.
Game-Changer Ahead: How Organ Age Tests Could Revolutionize Medicine
If these findings continue to hold up in larger clinical studies, they could reshape how medicine approaches aging and prevention.
Right now, doctors mostly wait for the disease to arrive and then treat it. A patient develops heart failure and gets medication. A patient shows signs of cognitive decline and gets tested for dementia.
What these studies describe is a different model entirely: measuring the biological age of specific organs from a routine blood draw, identifying which systems are aging fastest, and intervening before anything breaks.
Someone whose blood proteins suggest accelerated cardiovascular aging might start intensive heart-health interventions in their 40s rather than waiting for a diagnosis in their 60s. Someone with signs of immune aging might focus on anti-inflammatory strategies, sleep, and other factors known to slow that process.
The goal wouldn’t just be a longer life — it would be healthier years, with more function and less disease.
Caveats and Cautions: The Holes in Our Understanding of Aging
The researchers behind these studies are careful to point something out, and it’s worth taking seriously.
These are statistical models, not direct measurements of organ health. They identify correlations between protein levels and biological outcomes across large populations. They’re genuinely predictive, but they haven’t yet been validated as clinical tools — meaning your doctor can’t order an “organ age” blood test today.
The science is moving fast, though. Larger trials are underway, and some researchers believe tests like these could be in clinical use within a few years.
Paradigm Shift: Aging as Multiple Journeys, Not One Path
For most of human history, aging was something that happened to you, uniformly and inevitably.
What this research suggests is that aging is more like a collection of separate processes, running in different organs, at different speeds, some of which can be detected, tracked, and potentially slowed.
This is not a fringe idea from one research group. Looking beyond the Stanford papers, there are now more than 20 independent peer-reviewed studies from separate research teams around the world pointing in the same direction. Different methods, different populations, different labs, all arriving at a remarkably consistent conclusion: organs age at different rates, those differences are measurable, and they predict disease. When that many independent lines of evidence converge, it stops being a hypothesis and starts becoming a framework.
And the scientific community is already thinking about what comes next. If we can measure which organs are aging fastest, the logical next step is finding ways to slow them down, to keep each organ biologically younger for longer, and to prioritize intervention where it is most needed. Not a single pill. Not one universal treatment. Something far more personal than that.
This is a different way of thinking about health entirely. The goal is not simply to live longer. It is to protect the years that actually feel like living, to extend genuine health and function rather than just pushing the endpoint further out.
The question medicine has always asked is: how old are you?
The question it may soon be asking instead is: which part of you is aging fastest, and what are we going to do about it right now?
That shift, if it happens, would be one of the most significant changes in how we think about human health in a very long time—not reacting to the disease after it arrives. Anticipating it, organ by organ, years in advance, and choosing a different path before the damage is done.
And here is the part that should actually give you hope. You are not passive in this. The lifestyle choices you make every day, how you sleep, what you eat, how you move, and how you manage stress are already talking to your organs. Science is getting better at telling us which ones need the most attention. What we do with that information can make a bigger difference than most of us realize.
Watch this on YouTube
Sources:
Oh HS, Rutledge J, Wu J, et al. Organ aging signatures in the plasma proteome track health and disease. Nature. 2023;624:164–172. doi:10.1038/s41586-023-06802
Lehallier B, Gate D, Schaum N, et al. Undulating changes in human plasma proteome profiles across the lifespan. Nat Med. 2019;25(12):1843–1850. doi:10.1038/s41591-019-0673-2.
Oh HS, Lehallier B, Wyss-Coray T, et al. Plasma proteomics links brain and immune system aging with healthspan and longevity. Nat Med. 2025. doi:10.1038/s41591-025-03798-1.








Frustrated about steady decline in Thymus... the "finishing school" for immune system cells. There is some work under way but not widely recognized.
This really explains something you see all the time. Some people stay mentally razor sharp but their bodies slow down, while others remain physically active but start struggling cognitively. Good article!